Peptides constrained through multiple disulfides (or disulfide-rich
peptides, DRPs) have been an emerging frontier for ligand and drug
discovery. Such peptides have the potential to combine the binding
capability of biologics with the stability and bioavailability of
smaller molecules. However, DRPs with stable three-dimensional (3D)
structures are usually of natural origin or engineered from natural
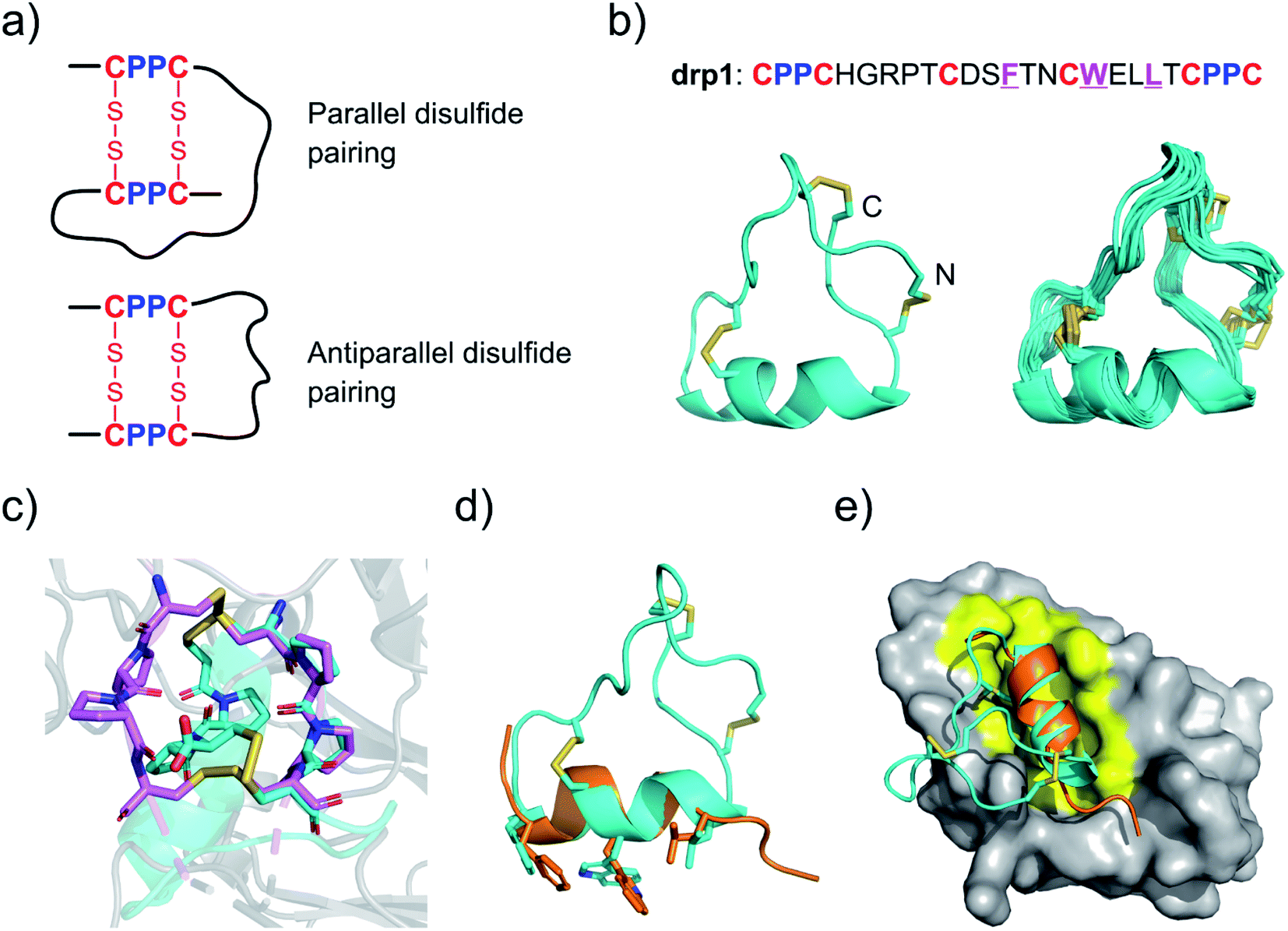
ones. Here, we report the discovery and identification of CPPC
(cysteine–proline–proline–cysteine) motif-directed DRPs with stable 3D
structures (i.e., CPPC–DRPs). A range of new CPPC–DRPs were
designed or selected from either random or structure–convergent peptide
libraries. Thus, for the first time we revealed that the CPPC–DRPs can
maintain diverse 3D structures by taking advantage of constraints from
unique dimeric CPPC mini-loops, including irregular structures and
regular α-helix and β-sheet folds. New CPPC–DRPs that can specifically
bind the receptors (CD28) on the cell surface were also successfully
discovered and identified using our DRP-discovery platform. Overall,
this study provides the basis for accessing an unconventional peptide
structure space previously inaccessible by natural DRPs and
computational designs, inspiring the development of new peptide ligands
and therapeutics.